Translational Perioperative and Pain Medicine (ISSN: 2330-4871)
ARTICLE DOI: 10.31480/2330-4871/167
Case Report | Volume 9 | Issue 4 Open Access
Anaesthetic Management in a Patient with Mitochondrial Encephalo Cardiomyopathy Undergoing a Dental Procedure: A Case Report
Denada Haka, MD*, Deniz Sivrioglu, MD, Nedim Cekmen, MD, PhD
Department of Anesthesiology, Faculty of Medicine, Baskent University, Ankara, Turkey
Denada Haka, MD, Department of Anesthesiology, Faculty of Medicine, Baskent University, Fevzi Cakmak Caddesi 10, Sokak No: 45 Bahcelievler, 06490 Ankara, Turkey, Tel: 0312203-68-68-4867; +905054349414, E-mail: denadahaka97@gmail.comEditor: Renyu Liu, MD, PhD, Professor, Department of Anesthesiology and Critical Care, Perelman School of Medicine at the University of Pennsylvania, Center of Penn Global Health Scholar, 336 John Morgan building, 3620 Hamilton Walk, Philadelphia, PA 19104, USA, Fax: 2153495078, E-mail: RenYu.Liu@pennmedicine.upenn.edu
Received: October 06, 2022 | Accepted: December 30, 2022 | Published: December 30, 2022
Citation: Haka D, Sivrioglu D, Cekmen N. Anaesthetic Management in a Patient with Mitochondrial Encephalo Cardiomyopathy Undergoing a Dental Procedure: A Case Report. Transl Perioper & Pain Med 2022; 9(4):499-502
Abstract
Mitochondrial myopathy (MM) is a rare syndrome caused by a defect in the mitochondrial DNA, resulting in defects in the formation of adenosine triphosphate (ATP) in the Krebs cycle, fatty acid oxidation and oxidative phosphorylation. From the anaesthesiologist's point of view, muscle fatigue and weakness, prolonged muscle blockage, hypothermia, tachycardia, acidosis and difficulty in breathing are some of the challenges to be faced during general anesthesia. In this case report, we present the case of a ten-years-old boy with TMEM 50 positive mutation undergoing dental procedures. We emphasize the importance of decreasing prolonged fasting ad hypoglycemia, postoperative nausea and vomiting, hypothermia, acidosis, and multidisciplinary assessment.
Keywords
Anesthesia management, Mitochondrial myopathy, TMEM 50, Lactic acidosis
Introduction
Mitochondrial myopathies are a group of rare pathologies caused by a mutation in the mitochondrial DNA. In these disorders, defects in the formation of adenosine triphosphate during the Krebs citric acid cycle, oxidative phosphorylation and fatty acid oxidation occur [1]. (MELAS) (mitochondrial encephalopathy, lactic acidosis, stroke-like symptoms) is a MM syndrome resulting in defects in the Krebs cycle's respiratory enzyme complexes I and IV, which are essential for normal aerobic metabolism [2]. The result is an imbalance between the energy requirements for multiple organs, including the heart, the central nervous system and skeletal muscles. There may be symptoms like heart failure, rhythm disturbances, seizures, visual and hearing problems, learning disabilities, autism spectrum disorder, muscle weakness and exercise intolerance. MELAS has an incidence of 12.5/100.000. TMEM 50 is one of the genetic mutations attributed to MELAS syndromes. From the anesthesiologist's point of view, exercise intolerance, muscle fatigue, muscle weakness, prolonged muscular blockage, tachycardia, hypothermia, and acidosis may cause concerns in high oxygen-demanding organs such as the brain, myocardium and muscles owing to disturbance of aerobic energy, metabolism caused by mitochondrial dysfunction. MELAS patients have an increased risk of lactic acidemia due to increased metabolic stress, as may be surgery or the appliance of anesthetic agents. There is no specific recommended procedure for these patients, and even though different anesthetic agents have been successfully used, severe complications may occur intraoperatively and postoperatively.
In this case report, we present the case of a ten-years-old boy with TMEM 50 positive mutation undergoing dental procedures.
Case Report
A 10-year-old boy with a body weight of 20.5 kg and average height compared to his age percentile of other children his age. His underlying disease was TMEM 50 mutation with concentric ventricle hypertrophy, light mitral regurgitation, hypospadias and an undescended testis anamnesis. The patient was on L-Carnitine, coenzyme Q10, vitamins C and E, riboflavin, and pregabalin treatment. He had a triangular face, droopy eyes and long limbs on inspection. His laboratory parameters were within the normal range. The patient was evaluated with Mallampaty Score II and American Society of Anesthetists III.
We obtained written informed consent from the patient's parents for the publication of this report. He was taken to the operating room where standard monitoring, including a pulse oximeter, electrocardiogram, capnography, noninvasive blood pressure, Bispectral Index (BIS) and Train of Four (TOF) were placed. After preoxygenation by 80% O2 for 3 min, general anesthesia was induced with propofol 2 mg/kg, fentanyl 1 μg/kg, and rocuronium 0.6 mg/kg. His mask ventilation was difficult because of his triangular face, so we used an oral airway to provide adequate ventilation. He had finger breadth oral excursion, average thyromental distance, normal upper and lower dentition, and standard neck extension and flexion. He was intubated with a size 5.5 cuffed endotracheal tube without any problem. Maintenance of general anesthesia was provided with a mixture of 2% sevoflurane and 50% oxygen in the air. An arterial blood sample obtained from the patient at the beginning of the operation showed lactic acidosis (pH: 7.29; lactate: 2.3 mmol/L, PaCO2: 45.3 mmHg, PaO2: 92.5 mmHg; HCO3: 21.1). He was given 500 mL of lactate free containing fluid intravenously (i.v.) to provide hydration. A circulating-water mattress and a forced-air warming device were used to prevent hypothermia. A glucometer measured blood glucose once in 30 minutes to avoid hypoglycemia. Hisglycemic level remained within the normal range during the operation. During surgery, the patient remained at 100% oxygen saturation, with expired carbon dioxide ranging from 35 to 40 mmHg, with a body temperature of 36.5 °C, adequate TOF 95% and BIS between 35-60 (Figure 1).
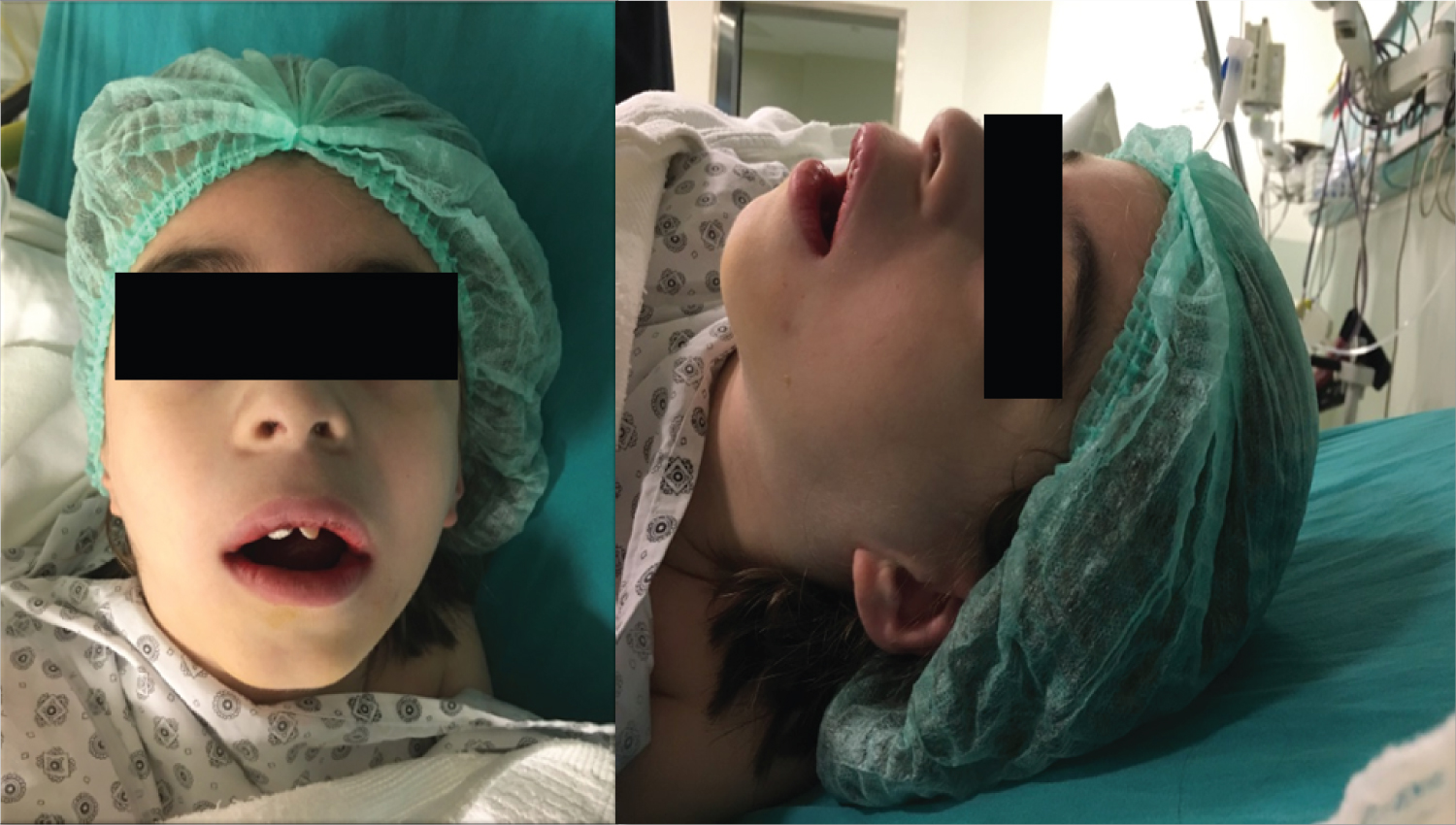
Figure 1: Patient during the intraoperative period.
The operation lasted 1 hour and 15 minutes. The patient was antagonized with 50 mg of sugammadex and extubated without complications. He was discharged from the operative room to the postoperative care unit and from the hospital one day later without complications.
Discussion
MELAS is a mitochondrial syndrome characterized by myopathy, encephalopathy, lactic acidosis, stroke-like episodes, diabetes and sensorineural hearing loss. Our patient had myopathy, encephalopathy and cardiopathy. The signs and symptoms of MELAS mostly appear during early childhood. Our patient was diagnosed with MELAS at the age of three due to muscle biopsy and DNA analyses. He had muscle weakness and pain, loss of appetite, attacks of vomiting and recurrent headaches due to continuously increased acidity in the blood. He had no history of stroke-like seizures. For preoperative evaluation in these patients, a head magnetic resonance, electromyography, echocardiography, BNP and exercise tolerance tests may be necessary.
All of the anesthetic agents potentially affect the mitochondria functions at different rates, and there is yet not a particular combination of drugs especially recommended for this group of patients. We used sevoflurane in our patient, which may cause more natural muscle relaxation than desflurane and isoflurane. Isoflurane and desflurane tend to depress the ventilatory response to CO2 more than sevoflurane. In mitochondrial myopathy, there may be an increased risk of malign hyperthermia triggered by volatile anesthetics, which is why we took protective measures to prevent malign hyperthermia when using sevoflurane. We prepared the machine by flushing it with oxygen at 10 l/min for ten minutes to reduce the anesthetic concentration to 1 part per million (ppm) or less. We used a nasal temperature probe for continuous temperature monitoring, TOF for proper muscle relaxation, and BİS for measuring the depth of anesthesia. In arterial blood gases obtained intraoperatively, electrolytes were recorded as underneath: Na 131, K 4.3, Ca 1.16, HCO3 21.1. At the end of the operation, an awake extubation procedure was applied, and no further complications occurred. We recommend performing these patients' surgeries in centers where Dantrolene medication is present in the operating theatre in case of an emergency.
Burnett B, et al. [3] used dexmedetomidine for anesthesia induction because they stated that the disruption of the electron transport chain and the Krebs cycle are at increased risk when propofol is used. In our patient, we preferred propofol because other agents like dexmedetomidine may precipitate hemodynamic instability at a higher rate [4,5]. In addition, no complications related to propofol usage as a bolus were recorded. It is the infusion of propofol that may result in propofol infusion syndrome in these patients.
Suzuki Y, et al. [6] used remimazolam bolus for induction and remimazolam infusion to maintain general anesthesia. We did not use benzodiazepine infusion because clearance is slow, and its effect may be prolongated.
MELAS patients have a more definitive risk of rhabdomyolysis and malignant hyperthermia than regular patients. The use of succinylcholine in MELAS patients with muscle wasting can still predispose patients to hyperkalemia; that is why we used rocuronium to provide neuromuscular relaxation and obtained arterial blood gases to detect the level of K+, which remained within average values.
None of the complications like myocardial disturbances, malign hyperthermia, or muscle hypotonia, occurred in our case. We decided to share this case report with our other colleagues around the globe as an essential source of information related to the role of general anesthesia because we could not find any other cases of patients with TMEM 50 undergoing general anesthesia [7].
In conclusion, meticulous preoperative planning should be done to minimize the metabolic stress caused by the process of anesthesia and surgery in these patients. We must remember that lactic acidosis and malign hyperthermia may develop in patients with MELAS. Pediatric patients with mitochondrial encephalo-cardiomyopathy should have their procedures done by trained multidisciplinary teams, including anesthesiologists, surgeons, pediatrists, and metabolic professionals at specialist centers. The importance of a perioperative comprehensive, detailed assessment and treatment for these patients is crucial.
Declaration of Interest
None.
Authors' Contribution
We took turns in caring for the patient. DH acquired the data and figures and produced the first draft of this report. DS analyzed the figures. NC reviewed and polished the manuscript.
Availability of Data and Material
The data generated or analyzed during this study are included in this article or, if absent, are available from the corresponding author upon reasonable request.
Acknowledgment
None.
Ethical Statement
Baskent University Hospital human research ethics committee approval was attended for writing this case report.
References
- Melonio C, Vieira C, Leal P,et al. Anesthesia for bariatric surgery in a patient with mitochondrial myopathy - case report. Braz J Anesthesiol. 2021;71(1):87-89.
- Kishida T, Ishida Y, Okada T, et al. Successful Perioperative Management of Cochlear Implantation in a Patient With Mitochondrial Encephalopathy, Lactic Acidosis, and Stroke-Like Episodes (MELAS). Cureus. 2022;14(8): e27761.
- Munlemvo D.M,. Syed A, Kimmet N. Anesthetic care for a child with undiagnosed myopathic/mitochondrial disease for gastrostomy tube placement and muscle biopsy.Pediatric Anesthesia and Critical Care Journal. 2020; 8(2): 133-139.
- Nelson KM, Patel GP, Hammond DA. Effects from continuous infusions of dexmedetomidine and propofol on hemodynamic stability in critically ill adult patients with septic shock. J Intensive Care Med 2020; 35: 875-880.
- Paliwal B, Rai P, Kamal M, Singariya G, Singhal M, Gupta P, Trivedi T, Chouhan DS. Comparison between Dexmedetomidine and Propofol with Validation of Bispectral Index For Sedation in Mechanically Ventilated Intensive Care Patients. J Clin Diagn Res. 2015 Jul; 9(7): UC01-5.
- Suzuki Y, Matsuyuki D, Yoshiki N. General anesthesia with remimazolam in a patient with mitochondrial encephalomyopathy: a case report. JA clinical reports. 2021;7:51.
- Brody KM. Anesthetic Management of the Patient with Mitochondrial Disease: A Review of Current Best Evidence. AANA J. 2022;90(2):148-154.PMID:35343897